 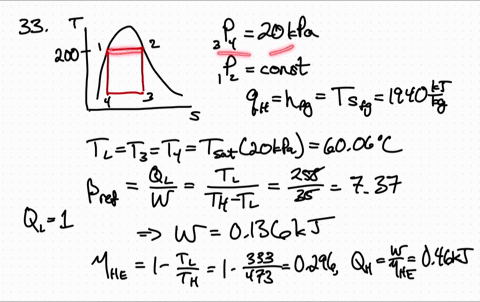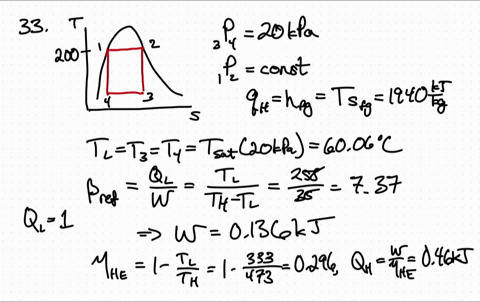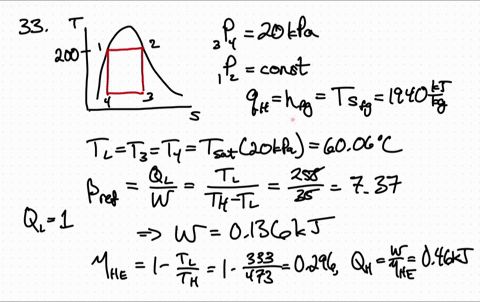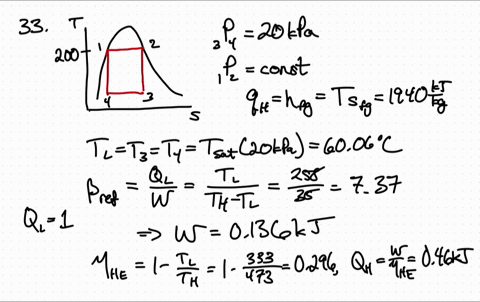 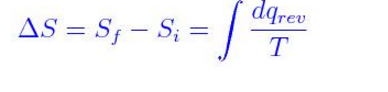
However, if the reaction occurs at high temperature the reaction becomes nonspontaneous, for the free energy change becomes positive when the high temperature is multiplied with a negative entropy as the enthalpy is not as large as the product. When the reaction occurs at a low temperature the free energy change is also negative, which means the reaction is spontaneous. When there is no item with label 1 in the set (p0) or if the set is full of items with Label 1 (p1), the entropy is zero. In 1934, Swiss physical chemist Werner Kuhn successfully derived a thermal equation of state for rubber molecules using Boltzmanns formula, which has since come to be known as the. The temperature would also determine the spontaneous nature of a reaction if both enthalpy and entropy were positive. Now have a look at the Entropy function, below. In short, the Boltzmann formula shows the relationship between entropy and the number of ways the atoms or molecules of a thermodynamic system can be arranged. Because both enthalpy and entropy are negative, the spontaneous nature varies with the temperature of the reaction. This equation can be derived from first principles of the properties of information. Using the entropy of formation data and the enthalpy of formation data, one can determine that the entropy of the reaction is -42.1 J/K and the enthalpy is -41.2 kJ. Equation for Entropy in a sample application for probability calculation: it is the sum over all values of a rv of the probability of that value times the log of that prob(i.e. Now one must find if the entropy is greater than zero to answer the question. Austrian physicist Ludwig Boltzmann explained entropy as the measure of the number of possible microscopic arrangements or states of individual atoms and. If the enthalpy is negative then the reaction is exothermic. 
for reversible process only Calculation of Entropy change. is an exact differential of some function which is identical as entropy 3. One may have to calculate the enthalpy of the reaction, but in this case it is given. has the same value irrespective of path as long as path is reversible 2. \): Matrix of Conditions Dictating Spontaneity Case 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
